

CBioProfiler(Cancer Biomarker and subtype Profiler)Platform
CBioExplorer is a tumor intelligent analysis platform, which mainly includes five modules: data dimensionality reduction, benchmark experiment, predictive model construction, and clinical and biological annotation of biomarkers. It can achieve fully automated screening, validation, and analysis evaluation of clinical tumor markers to ultimately guide clinical decision-making. The platform supports the data selection of TCGA, GEO, ICGC, Array-Express, CGGA and other international public libraries, and also supports the use of user's own data. And it integrates the six frontier model of machine learning based on survival analysis and new R language package (CuratedCancerPrognosisData), to achieve cancer survival related biomarkers from molecular level research to the transformation of the clinical application. The visual interface, user-friendly and easy to operate, will provide important assistance for clinicians and relevant researchers to facilitate data analysis and clinical transformation applications.
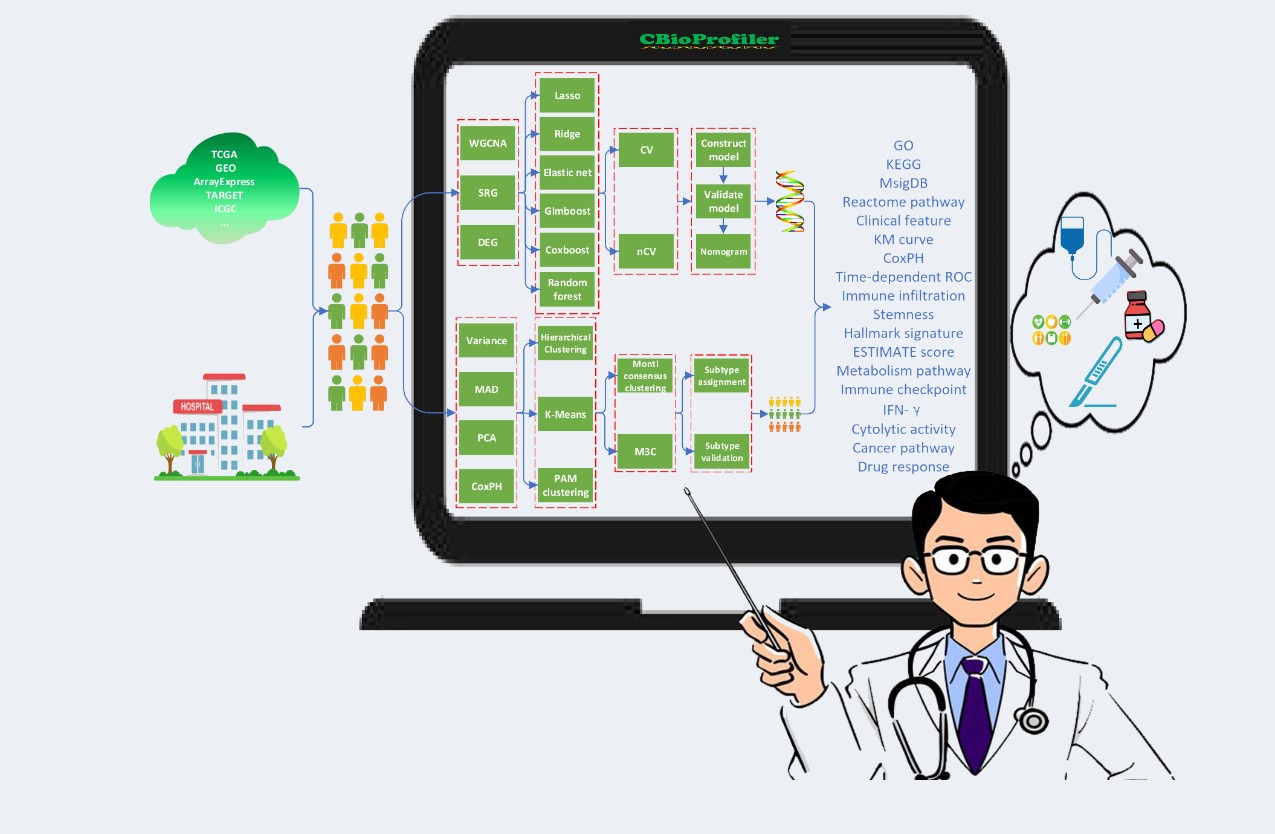
Access link: https://cbioprofiler.znhospital.cn/CBioProfiler/